THE EFFECT OF SHORT-TERM WEIGHT-BEARING EXERCISE ON BONE MASS DENSITY IN OBESE AND THIN YOUNG GIRLS
Habibzadeh Nasim
Department of Sport Sciences, Exercise Physiology, University of Guilan, Rasht, Iran.
Original scientific paper
Abstract
The purpose of the present study was to examine the effect of walking program on bone mass density (BMD) among healthy obese and thin young girls. Twenty untrained obese (n=10) and thin (n=10) girls, 20-25 years, volunteered to participate in this study. Before and after the training program both groups had anthropometric measurements, blood analysis BMD evaluation. Each walking session was 30 minutes walking between 50-75% of maximal heart rate, 3 days per week for 2 months. After exercise program, BMD in both regions (hip (1.1%), spin (L2– L4) (2.3%)) stabilized in the both groups (p<0.05). Percent body fat, fat mass and lean mass were affected positively by exercise program (all p = 0.000). No significant change was observed in Serum estrogen calcium, phosphorus in either group (p>0.05). This study showed that activities such as walking provide significant loading, which positively influences BMD in young thin and obese girls. This result suggests that both thin and obese women can reduce the risk of bone loss by increasing their level of activity.
Key words: Bone mass density, Bone loss, Walking exercise, Obese, Thin.
INTRODUCTION
Osteoporosis is a major public health problem that is characterized by low bone mass and increased susceptibility to fracture, primarily in the hip, spine and wrist (1). Although symptoms of osteoporosis do not generally occur until after menopause, recent evidence suggests that bone loss starts much earlier in life and it may be associated with an increasingly sedentary lifestyle (2). That is why the world Health Organization believes that we are heading for a major epidemic the years to come. In spite of development in diagnosis of osteoporosis, still the preventive measure of osteoporosis is neglected and the already staggering medical, social and economic costs can be expected to increase unless effective prophylactic and therapeutic regimens are developed (3).
Body weight impacts on bone density and therefore is an important risk factor for osteoporosis (4). It is well recognized that thin individual have lower bone mass density than heavier individual and there may be multiple reason for this. In fact thinness is an important risk factor for bone loss and a reduction in bone mass is highly correlated with an increased of osteoporosis (5) . Obesity has been identified as a risk factor many illnesses. The consequence of excessive weight can have a profound negative effect on bones and joints. An increased body mass index (BMI) has been associated with many orthopedic conditions, such as arthritis, osteoporosis, and joint immobility (6).
To prevent osteoporosis various factors such as physical activity, adequate ingestion of calcium and vitamin D are acknowledged to be important (7). Because mechanical loading contributes to subsequent bone mass, weight–bearing exercise is suggested as a therapy to increase BMD and as a strategy for preventing osteoporosis (8). Numerous studies demonstrate the importance of weight–bearing physical activity as well as mechanical loading for maintaining skeletal integrity in both younger and elderly women. Lack of weight-bearing activity is dangerous for skeletal and a decrease in bone mass has been demonstrated in humans under condition of weightlessness or immobilization (9). Walking is a weight-bearing from of aerobic exercise that can be easily integrated into one' s daily life and it is frequently recommended as a way to help protect against bone loss(10).While various forms of weight-bearing activity may slow loss of BMD or possibly increase BMD through mechanical loading of bone, walking as an exercise intervention is of particular interest (11). Several cross-sectional and descriptive studies indicate a positive relationship between walking and BMD at various skeletal sites. Whereas, other studies have shown that walking was insufficient for improving BMD (12).
It is known that the skeletal response to loading is characteristic of different age (13). However to our knowledge, no study has shown the effect of exercise program on BMD in obese and thin girls with a mean age 20 years simultaneously. Clearly, further studies are required to determine whether exercise programs which are acceptable to older people are effective in preventing osteoporosis in this population. Therefore the purpose of the present study was to examine the effect of walking program on hip and lumbar (L2 – L4) BMD in order to prevent or restore bone loss among healthy sedentary obese and thin girls.
METHODS
Subjects
Twenty young physically untrained volunteered to participate in this study. Then participant were pair wise -matched and assigned to two exercise obese and thin groups. Descriptive statistics of the subjects' anthropometric and physiological characteristics are presented in table 1. Written informed consent for all procedures was obtained from all participants prior to entering the study. The criteria for the invitation were being willing to participate, clinically healthy (no cardiovascular, musculoskeletal, respiratory, or other chronic diseases that might limit training or testing), no menstrual irregularities, not using medication that alert bone mass density and no beta-blockers, sedentary life style (no regular sports activities for at least 2 years), nondieting, nonsmoking, and no apparent occupational or leisure time responsibilities that impede their participation.
Table1.
Physical characteristics of study subjects(X ±SD)
Thin (n=10) |
Obese(n=10) |
Variable |
21.10 ± 1.7 |
22.22 ± 1.9 |
Age(year) |
159.90 ± 7.5 |
5.1 ± 157.78 |
Height (cm) |
45.88 ± 5.3 |
74.98 ± 8.1 |
Weight (kg) |
17.73 ± 1.0 |
30.20 ± 1.8 |
BMI (kg/m2) |
Anthropometric measurement
Anthropometric and body parameters were assessed after participants had fasted overnight.
Height was measured to the nearest 0.1 cm using a wall-mounted stadiometer (Holtain, Dyfed, UK). Body mass was measured to the nearest 0.01 kg on an electronic weighing scale (Mettler Toledo IDL Plus, Eichfahig, Germany) .Body mass index (BMI) was calculated as weight (kg)height (m2). The In addition, all subjects were weighed every week so that none of them gained or lost > 2.2 kg body weight over the entire study period.
BMD assessment
The main endpoints of the study were the change in bone mass density of the hip and the lumbar spine (L2 – L4).BMD (g/cm2) was also measured with the dual X-ray absorptiometry scans (DXA) (Lunar DPX-L, software version 1.31, USA). All the scanning and analyses were done by the same operator. The vivo day-to-day (coefficient of variation) of the BMD measurement in our laboratory range from 0.7 to 1.7%.The scanner was calibrated daily, and its performance was followed with our quality assurance protocol. There was no significant machine drift during the study period.
Blood collection and analysis
Blood samples were collected after an overnight fast (>12 h) in a sitting position and centrifuged at 1500 rpm for 30 minutes at 4o C within 2 h. Serum samples from each participant were stored frozen at -20o C until analyzed. Serum estrogen level was assessed by radioimmunoassay (Amersham Biosciences, Piscataway, NJ, USA) in follicular stage in each subject's menstrual cycle and serum calcium, phosphorus levels were measured by standard automated laboratory techniques.
Dietary Intake
Caloric expenditure was calculated based on the weight of the subject. To minimize any affect that dietary composition might have on the measured metabolic variables, the initiation of the study all subjects were instructed on the American Health Association (AHA) diet by registered dietitian. The composition of this diet was 50-55% carbohydrate, 15-20% protein, <30% fat. The subjects were asked to maintain this diet composition throughout the study's duration (2mo).Compliance was monitored by review of 7-day food records taken every week (14).
Exercise program
The exercise program included warming-up phase for 5 minutes of stretching exercises, 30 minutes walking at 50-75% of maximum heart rate and cooling-down phase for 5 minutes of stretching, three times a week for 2 months. Stretching exercises were performed for the arms, leg, back and stomach. A target heart rate range between 50-75% of age adjusted maximum heart rate intensity was calculated by each walker from her age and walking supine resting heart rate in every exercise session (15). Heart rate was measured with an electronic heart rate meter (Sport Tester PE, Polar Electro, Oy, Finland). The exercise program was accompanied by music. All sessions were supervised by a professional exercise physiologist leader.
Statistical Analysis
Mean and standard deviation (SD) was used as descriptive statistic. Student's t-test was used for normally distributed variables. Unpaired t-test was used to assess the change in BMI, body weight, serum calcium, phosphorus, and estrogen before and after the exercise intervention. The effect of the program was studied through a mixed, two-factor analysis of variance(2×2) incorporating the group, time and group –by-time interaction effect. A significance level of set at p<0.05 was used for all comparisons.
RESULTS
All twenty subjects (100%) completed the training program. No major change in menstrual status was observed during the study. All subjects showed normal ranges of serum calcium, phosphorus, or estrogen levels at the baseline and analysis of data showed that the post-test differences between the groups were not significant (p > 0.05).
Percent body fat, fat mass and lean mass changes in response to training were significant in the two groups. The lean mass in both groups were significantly increased but the present body fat, fat mass were significantly decreased (p = 0.000).
The BMD values of the spin (L2– L4) and hip were both significantly different between the two groups and exercise significantly increased the BMD from baseline (p<0.05).The comparison of the changes in BMD at the hip(1.1%), the bone mass density at the spin (L2– L4)(2.3%)showed a trend for higher effectiveness of the exercise. The different effects observed at the two sites studied could be attributed to one important factor. The lumbar vertebrae are 65% trabeculare bone, one characterization of which is grater metabolic activity. The hip, on the other hand, is 75% cortical bone, which is more compact and less quickly reactive to treatment (Lawence et al, 2005). However, both spinal and hip BMD, were affected positively by the exercise program. Figures 1, 2 show the change in hip and spinal (L2– L4) BMD over the study period and the significance of difference in the both thin and obese groups.
Table 2.
Change in variables (X ±SD)
variable |
Thin |
Obese |
P value |
||
pretest |
posttest |
pretest |
posttest |
||
Hip BMD |
.843 ± 0.05 |
0.863 ± 0.06 |
.967 ± 0.10 |
.983 ± 0.09 |
0.012* |
Spine |
1.051 ± 0.14 |
1.128 ± 0.21 |
1.113 ± 0.16 |
1.147 ± 0.15 |
0.037* |
Estrogen |
25.55 ± 8.93 |
42.15 ± 18.80 |
30.42 ± 15.60 |
46.99 ± 18.55 |
0.610 |
Calcium |
9.78 ± 0.42 |
9.25 ± 0.50 |
9.47 ± 0.24 |
9.42 ± 0.28 |
0.783 |
Phosphorous (ml/dl) |
4.21 ± 0.39 |
3.55 ± 0.36 |
3.80 ± 0.39 |
3.65 ± 0.63 |
0.660 |
Lean mass |
33.54 ± 3.72 |
34.53 ± 3.97 |
43.27 ± 5.25 |
44.38 ± 6.21 |
0.000* |
Fat mass |
9.86 ± 1.85 |
9.21 ± 2.14 |
29.11 ± 4.54 |
27.17 ± 6.30 |
0.000* |
% Body fat |
21.82 ± 3.13 |
20.13 ± 3.60 |
38.80 ± 3.97 |
36.35 ± 6.84 |
0.000* |
* Significantly different from the 'Pre' value:* p < 0.05;***
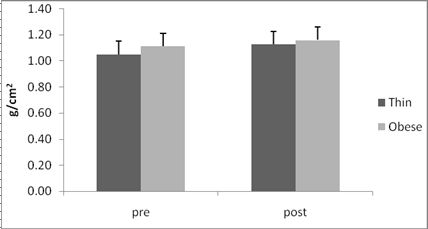
Fig1.Change from baseline in Spine (L2– L4) BMD (g/cm2) during the study period
(Pre, post study) and the significance of the difference between the groups ANOVA (p=0.037).
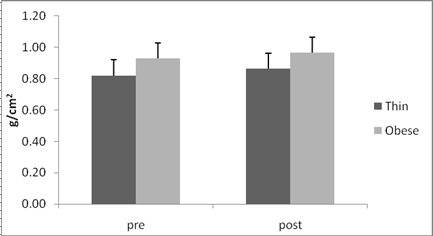
Fig2. Change from baseline in Hip BMD (g/cm2) during the study period
(Pre, post study) and the significance of the difference between the groups ANOVA (p=0.012).
Discussion
This is the first study to assess the effect of weight-bearing exercise on bone loss at different anatomical sites in different young girls who are considered to be at risk of osteoporosis. Two months of walking exercise showed efficacy in slowing or stopping bone loss. This difference may be due to the younger age of subjects in our study and the nature of the mechanical load. Furthermore, there was a relation between weight change and increase in BMD in both thin and obese girls.
Exercise will be part of an effective strategy to reduce the incidence of osteoporosis only if the amount and type of physical activity needed to confer benefit is attainable for majority of women. The most easily accessible from of weight-bearing exercise is walking and the number of hours of walking per day has been founded to be correlated with lumbar and hip densities (16).
The minimum weight-bearing exercise recommended for prevention of osteoporosis in half an hour three times a week (17) , however all exercise are not good for the human body, therefore the degree and the extent of any exercise should be adapted to the age, the physical ability , the skeletal condition of the individual. Comparisons among studies evaluating the effect of walking on bone density at various skeletal sites are limited by differences in methods to measure walking activity and differences in menses and nutritional status of the study population. For example Cavnaugh and Cann have reported that aerobic exercise such as walking program did not prevent bone loss(18).Hotori et al also reported that walking for 30 minutes above the anaerobic threshold (AT) was effective in increasing BMD, whereas exercise below the AT was not(19).Martin and Notelovitz similarly observed that walking speeds of less than 6.4 km did not increase BMD(20).Other studies have shown that physical exercise positively affected BMD in both young and elderly women. Among postmenopausal women, Nelson showed the beneficial effects on BMD of walking at 75-80% of maximum heart rate for 50 minutes, 3-4 times per week, wearing a leaded belt (21).Yamazaki et al (2004) also demonstrated that the positive effect of 1 year of moderate walking exercise on the BMD was caused by a decrease in bone turnover in postmenopausal women (22). These finding confirm that exercise decreased bone turnover, which was elevated by estrogen deficiency, and resulted in positive effect the skeleton. A recent study by Mulhim et al has shown that 30 minutes of walking at 1.5km/hour was increased lumbar spine and femoral BMD in sedentary Saudi woman, aged 25-50 year (23). They suggested that the bone maintenance effect of exercise during the premenopausal and postmenopausal period may be an essential factor, making a favourable difference as compared with sedentary women. Brooke-Wavell et al also reported that 20 min walking a day increased BMD (24).
In our patient a simple 30 minutes of walking exercise at the range of 50-75% maximum heart rate was enough to positive change the BMD, deceased the fat mass and increase in lean body weight, making them healthier. Mechanical factors that affect bone remodelling include muscular contraction and gravity. Lanyon found that bone responds in proportion o the amount of stress place on it. Abramson and Dwlagi showed that weight bearing and muscle contractions generate stress on bone necessary to prevent bone loss (25).
Although overall fat mass does improve bone density, so does overall lean mass. “Lean mass” means muscle."Lean mass," the researchers conclude, "is the major determinant of bone size, providing further evidence that bone size is adapted to the dynamic load imposed by muscle force rather than passive loading" by fat(26). Result of the blood parameters showed that neither estrogen nor calcium and phosphorous levels were significantly altered as a result of two-month training regimen, suggesting that estrogen, calcium and phosphorous did not mediate the observed skeletal changes in the both groups.
In conclusion, our study showed that activities such as walking provide significant loading, which positively influences BMD in sedentary young girls. This result suggests that both thin and obese women can reduce the risk of bone loss by increasing their level of activity. If done on a regular basis, this type of training can be efficient, safe an inexpensive way of preventing osteoporosis and osteoporosis related fractures later in life.
Acknowledgment
The work was supported by grants from the University of Guilan. The authors gratefully acknowledge the all subjects whom cooperated in this investigation.
REFERENCES
1.Abdy SE. A strategy to prevent osteoporosis. Prac Nurse 1998:16:614-619.
2.Hata M, Miyom M, Mizuno Y. Osteoporosis as a lifestyle related disease. Nippon Rinsho 2003: 61:305-313.
3. Sambrook P, Cooper C. Osteoporosis. Lancet 2006 :367:2010-18.
4. Douchi T, Oki T, Nakamura S, Ijuin H, Yamamoto S, Nagata Y. The effect of body composition on bone density in pre-and postmenopausal women. Maturitas 1997:27:55-60.
5. Coin A, Sergi G, Beninca P, Lupoli L, Cinti G. Bone mineral density and body composition in underweight and normal elderly subjects. Osteoporos Int 2000:11:1043-50.
6. Cobayashi F, lopes L, Taddei J. Bone mineral density in overweight and obese adolescents. Journal Pediatria 2005: 81:337-420.
7. Renfro J, Brown JB. Understanding and preventing osteoporosis. AAOHN J 1998: 46:181-191.
8. Turnerc H. Exercise as a therapy for osteoporosis. Bone 1998:23:83-85.
9. Alioa JF, Cohn SH, Ostuni JA, Cane R, Ellis K. Prevention bone loss by exercise. Ann Intern Med 1987 :89:356-8.
10. Palombaro KM. Effect of walking-only interventions on bone mineral density at various skeletal sites: a meta-analysis. J Geritr Phys Ther 2005:28:102-107.
11. Mosisio KC, Hurwitz DE, Sumner DR. Dynamic loads are determinates of peak bone mass. J of Orthopedic Research 2004:22:339-345.
12. Berard A, Bravo G, Gauthier P. Meta-analysis of the effectiveness of physical activity for prevention of bone loss in postmenopausal women. Osteoporos Int 1997:7:331-337.
13. Iwamoto J, Takeda T, Ichimura S. Effect of exercise training and detraining on bone mineral density in postmenopausal women with osteoporosis. J Orthop Scin 2001:6:128-132.
14. New SA, Smith C, Grubb DA, Reid DM. Nutritional influence on bone mineral density: a cross sectional study in premenopausal women. AM J Clin Nutr 1997: 65:1831-9.
15. Karvaonen MJ, Vuorimaa T. Heart rate and exercise intensity during sport activities: Practical application. Sport Medicine 1988:5: 303-312.
16. Elizabeth K, Bess H. Walking is related to bone density and rates of bone loss. The American J of Medicine 1994:96:20-26.
17. Kato Y, Takata KI, Yasaku K, Koitaya N. Walking during and habitual exercise related to bone mineral density using computer-assisted X-ray densitometry in Japanese women. Geriatric and Gerontology Int 2005: 5: 176-181.
18. Cavanaugh DJ, Cann CE. Brisk walking did not stop bone loss in postmenopausal women. Bone 1988: 9:201-204.
19. Hatori M, Hasegawa A, Adachi H, Shynozaki A, Hayashi R, Okano H. The effects of walking at the anaerobic threshold level on vertebral bone loss in postmenopausal women. Calcif Tissue Int 1993:52:411-414.
20. Martin D, Notelovize M. Effects of aerobic training on bone mineral density of postmenopausal women. Journal bone Mineral Research 1993: 8:931-936.
21. Nelson Miriam E, Fisher Elizabeth C, Dilmaniam F Avraham A 1-y walking program and increased dietary calcium in postmenopausal women: effect on bone. Am J Clin Nutr 1991:53:1304-11.
22. Yamazaki S, Ichimura S, Iwamato Jun, Takeda T, Totama Y. Effect of walking exercise on bone metabolism in postmenopausal women with osteopenia /osteoporosis. J Bone Miner Metab 2004 :22:500-508.
23. Mulhim AA, AL Gazzar S, AL Bahnassy A, Al Mir S. Effect of exercise program on bone mineral density in sedentary females. Bahrain Medical Bulletin 2004: 26:1-5.
24. Brooke Wavell K, Jones PM, Hardman AE, Tsuritani I, Yamada Y. Commencing, continuing and stopping brisk walking: effects on bone mineral density, quantitative ultrasound of bone and markers of bone metabolism in postmenopausal women. Osteoporos Int 2001:12:581-587.
25. Abramson AS, Delagi EF. Influence weight bearing and muscle contraction on disuse osteoporosis. Arch Phys Med Reabil 1961:42:147-51.
26. Kaufman JM. Muscle mass, not fat, makes for stronger bones. Journal of Clinical Endocrinology and Metabolism 2009.www.canada.com/health.
EFEKTI KRATKOTRAJNOG VJEŽBANJA SA DODATNIM TEGOVIMA NA KOŠTANU MASU KOD PRETILIH I MRŠAVIH DJEVOJAKA
Originalni naučni rad
Sažetak
Cilj istraživanja je bio da se istraže efekti programa pješačenja na gustinu kostiju (BMD) kod zdravih pretilih i mršavih djevojaka. Dvadeset djvojčica koje nisu u trenažnom procesu, pretilih (n=10) i mršavih (n=10), 20-25 godina starosti, su dobrovoljno učestvovale u istraživanju. Prije i poslije trenažnog programa objema grupama su izmjerene antropometrijske karakteristike, te urađena analiza krvi i gustine koštane mase. Svaki trening pješačenja je trajao 30 minute u rasponu od 50-75% maksimalnog pulsa. Program je trajao 2 mjeseca, tri puta sedmično. Nakon sprovedenog programa, koštana masa se stabilizovala u obe regije (kuk, 1.1 % i lumbalni dio kičme 2.3%) na statistički značajnom nivou (p<0.05). Program vježbanja je pozitivno uticao na procenat masnog tkiva u tijelu i njenu masu, kao i na masu tijela oslobođenu masnog tkiva (p=0.00). Statistički značajne promjene nisu pronađene u visini nivoa serum estrogena, calcijuma i fosfora. Ovo istraživanje je pokazalo da aktivnost kao što je šetnja osigurava dovoljno opterećenje, koje pozitivno utiče na gustinu koštane mase kod pretilih i mršavih djevojaka. Rezultati sugerišu da i pretile i mršave djevojke mogu prevenirati rizik od gubitka koštane mase povećavajući nivo aktivnosti.
Ključne riječi: gustina koštane mase, gubitak koštane mase, pješačenje, pretilost, pothranjenost
Correspondence to:
Habibzadeh Nasim, MSc
Department of Sport Sciences, Exercise Physiology,
University of Guilan, Rasht, Iran.
Tel: 09111376241
Email: nasim_habibzadeh@yahoo.com